Data publication for personalized health data as new standard
NFDI4Health creates procedures for FAIR publication of health studies without compromising data protection.
Publication of study data
NFDI4Health has established a metadata standard and process for the publication of health studies to make health data FAIR:
- Based on existing international standards, NFDI4Health has developed a generic information model for describing health-related studies and is in communication with various groups to establish this information model as a national and international standard. Read more. Read more about the implementation guideline of the information model.
- This information model also includes information on data sharing possibilities and the corresponding access conditions. Together with a persistent identifier, all (meta)data are thus published in order to speak of a data publication. The data itself remains under the full control of its primary custodians and access can be requested through the established application procedures. Read more about the publication guidelines and the metadata schema.
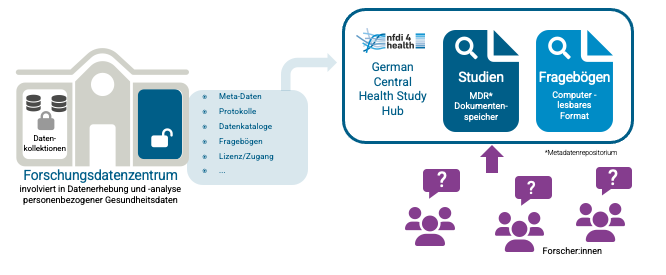
- For health study publication, NFDI4Health has developed the German Central Health Study Hub as an infrastructure. It allows study metadata to be collected via a user-friendly web-based data collection template or application programming interface. The German Central Health Study Hub also provides the ability to publish study documents individually, with the publication of questionnaires and variable catalogues being particularly desirable, as these contain the most detailed information about available data. In this way, health studies with associated data become more discoverable, and researchers can use the information to evaluate the content of data collections and learn about access conditions and requirements for the data. Read more.
Health Study Hub
The German Central Health Study Hub allows researchers to publish their project characteristics, documents and data related to their research project in a FAIR manner or to find information about past and ongoing studies.
Data Train
The Data Train cross-disciplinary graduate training programme, a core element of the NFDI4Health training approach, aims at building the next generation of data-savvy researchers in the biomedical sciences.
Personal Health Train
Local Data Hub
Data publication
Health data, as collected in clinical trials and epidemiological, as well as public health studies, cannot be freely published, but are valuable datasets whose reuse is of high importance for health research. NFDI4Health has established a metadata standard and process for the publication of health studies to make health data FAIR.
 Deutsch
Deutsch 