Our offering for researchers
NFDI4Health provides a technical infrastructure and efficient services for FAIR health data. The aim of these services is to make data from health research findable and accessible, while ensuring data protection and adherence to ethical standards. Users are also supported through a wide range of support and training services.
Health data, as collected in clinical and epidemiological studies as well as public health studies, are extremely valuable datasets for health research. They enable the improvement of the health of all people. At the same time, personal health data are particularly sensitive and in need of protection, which complicates their free publication.
NFDI4Health develops standards, services, and infrastructure solutions that make health data sustainably and FAIR (Findable, Accessible, Interoperable, Reusable) for research, also in exchange with other national and international health research data infrastructures. An important aspect is the cooperation with other NFDI consortia across domain boundaries.
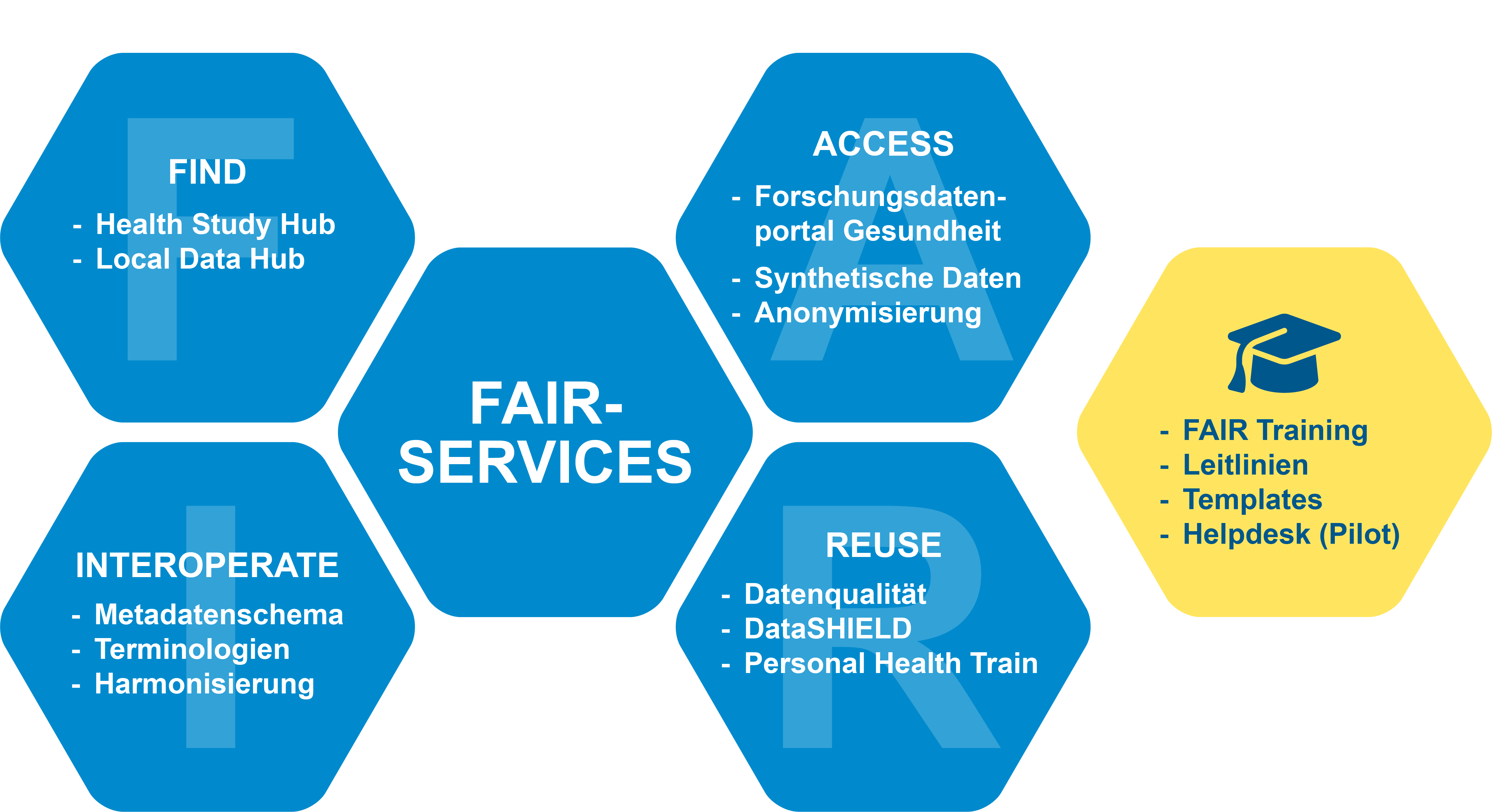

Health Study Hub
The Health Study Hub enables the publication and discoverability of clinical, epidemiological, and public health data, and promotes their reuse. Users can narrow their searches to specific metadata, instruments, and other resources. In addition, variables from individual datasets can be compared with one another.
Local Data Hub
The Local Data Hub is a software tool for local and standardised data management in health research. This decentralised registry helps researchers organise and share their studies, datasets, and protocols. Through an interface, the information can be published in the Health Study Hub, making it globally discoverable.

Portal for Medical Research Data (FDPG)
Through the German Portal for Medical Research Data (FDPG) of the Medical Informatics Initiative (MII), healthcare data can be requested via a standardised process. NFDI4Health is currently expanding the portal’s functions in close collaboration with the MII and the Network University Medicine (NUM), so that clinical, epidemiological, and public health data can also be centrally requested. The provision of data is centrally coordinated and made transparent via the project registry.
Synthetic data
Synthetic data replicate the structure of real health data, thereby creating a data protection-compliant foundation for research and analysis. NFDI4Health supports researchers with appropriate methods and tools for the generation, validation, and visualisation of such data.
Anonymisation
Anonymisation enables the secure sharing of sensitive data while complying with legal requirements. NFDI4Health provides advice and support in the generation, evaluation, and risk assessment of anonymised data. The anonymisation software ARX supports a wide range of data protection and risk models, as well as data transformation methods that minimise the risk of re-identification.

Metadata schema
Metadata describe and organise data, making them easier to find and reuse. The NFDI4Health metadata schema captures and describes metadata from health research and forms the basis for machine-readable, standardised publication in the Health Study Hub. A flexible modular system allows for use across various areas of health research.
Terminologies
Terminologies encompass all specialised terms within a field and define globally standardised concepts. NFDI4Health provides a terminology service that enables users to access standardised terms and metadata from health research. With the Annotation Workbench, researchers can categorise their data and assign standardised codes.
Harmonisation
As the collection and documentation of research data have evolved over time and often differ between studies, these datasets are not always directly compatible. To enable cross-study analysis of existing research data, NFDI4Health provides a harmonisation strategy.

Data quality
High data quality is a cornerstone of health research, enabling optimal data reuse. NFDI4Health offers dataquieR, an R package for assessing the quality of health data, and continues to develop it based on users’ needs.
DataSHIELD
DataSHIELD is software for privacy-preserving distributed data analysis. A secure processing environment is created, allowing distributed data to be analysed in a decentralised and secure manner. NFDI4Health is currently extending DataSHIELD specifically for the analysis of clinical, epidemiological, and public health data, and supports data holders in setting up and using the software.
Personal Health Train
Personal Health Train is another software tool for secure and privacy-compliant distributed analysis and is suitable for a wide range of health data. Here too, a secure processing environment is established, enabling decentralised and safe analysis of distributed data. The software is currently in the pilot phase and is being tested by NFDI4Health for image analysis.

FAIR Training
The NFDI4Health FAIR Training offers both online and in-person courses on research data management. The programme trains researchers at all career stages in the FAIR principles and their application across the various phases of the data lifecycle. All training materials are freely available and are supplemented with resources for trainers.
Guidelines
NFDI4Health defines clear standards in its guidelines and outlines best practices in health research. All materials are available for free download. Currently, available resources include a publication guideline, a training handbook, a white paper on record linkage, and templates for data management plans.
Helpdesk
Our Helpdesk is the point of contact for all questions – whether related to organisational matters, the management of personal health data from clinical, epidemiological, or public health research, or regarding our tools and services.
 Deutsch
Deutsch 