Was ist die Idee hinter NFDI4Health?
-
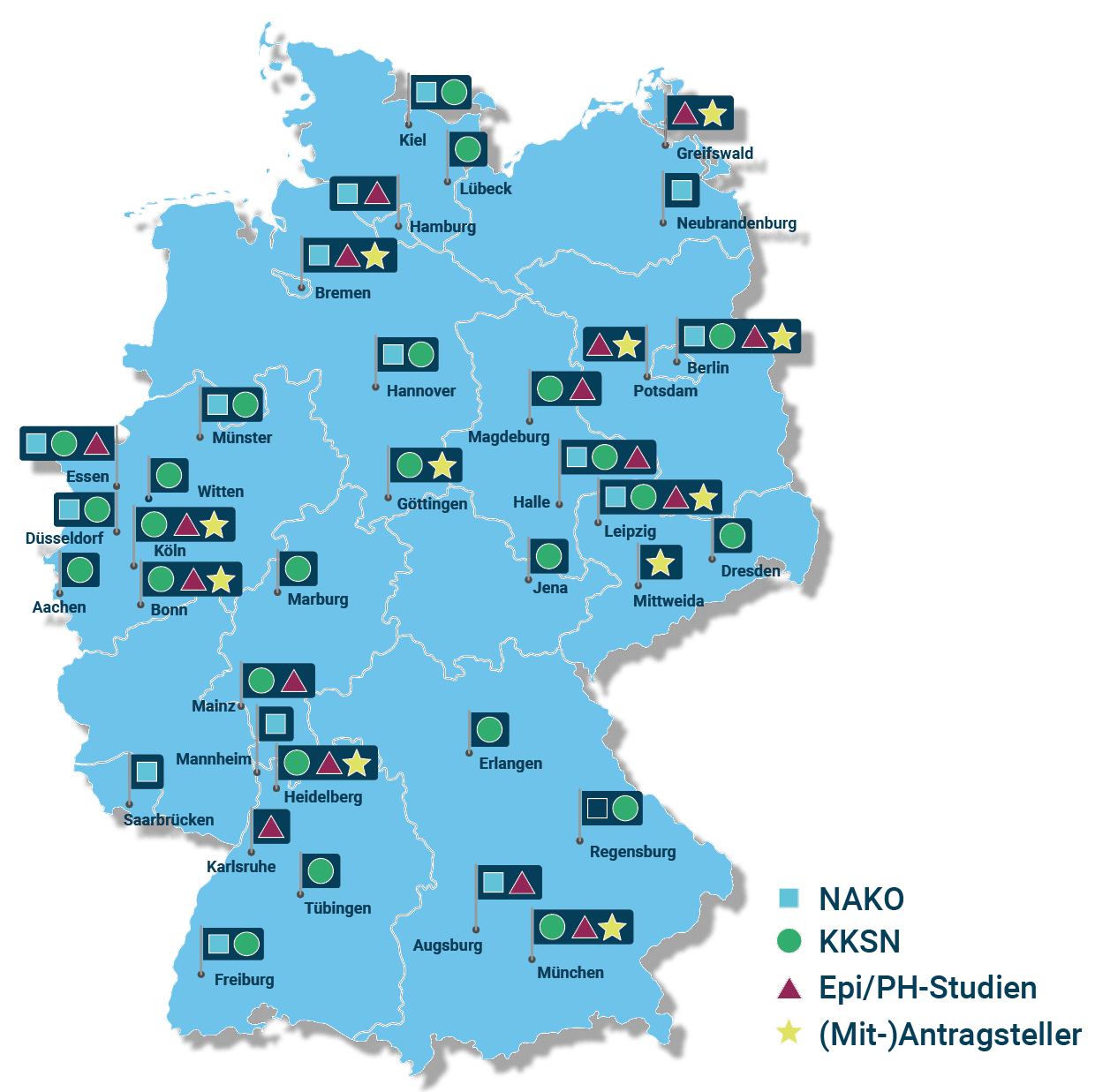
Umfassende Abdeckung epidemiologischer und klinischer Studien sowie der Public Health-Forschung mit hoher Datenqualität in Deutschland
-
Starke Einbindung der Nutzercommunity bei der Entwicklung von NFDI4Health
-
Besondere Kompetenzen des Konsortiums: Langjährige Erfahrung in den Bereichen Forschungsdatenmanagement sowie der Aus- und Weiterbildung; Dateninhaber mit hohen IT-Standards, etablierten Nutzungs- und Zugriffsverfahren, hohen Datenschutzstandards; transdisziplinäre Zusammenarbeit von Kliniker:innen, Epidemiolog:innen, Jurist:innen, (biomedizinischen) Informatiker:innen, Ernährungswissenschaftler:innen, Statistiker:innen
-
Eingebunden in ein breites Netzwerk von (inter-)nationalen Initiativen / Organisationen
-
Hervorragend aufgestellt, um eine nachhaltige Infrastruktur für Datenaustausch zu schaffen
 Deutsch
Deutsch  English
English